 Let’s revisit this old rule, and put our knowledge of covalent and noncovalent bonding to work. 
You probably remember the rule you learned in general chemistry regarding solubility: ‘like dissolves like’ (and even before you took any chemistry at all, you probably observed at some point in your life that oil does not mix with water). It is critical for any organic chemist to understand the factors which are involved in the solubility of different molecules in different solvents. 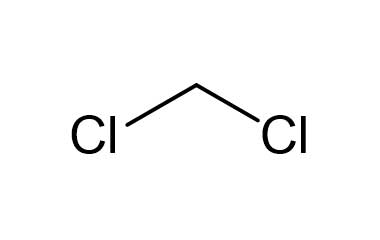
In organic reactions that occur in the cytosolic region of a cell, the solvent is of course water. In recent years, much effort has been made to adapt reaction conditions to allow for the use of ‘greener’ (in other words, more environmentally friendly) solvents such as water or ethanol, which are polar and capable of hydrogen bonding. In the organic laboratory, reactions are often run in nonpolar or slightly polar solvents such as toluene (methylbenzene), hexane, dichloromethane, or diethylether. Virtually all of the organic chemistry that you will see in this course takes place in the solution phase. 99%) + primary bromide, trace amount” width=”477″ height=”153″> Figure 9.4f An example of the bromination of isobutaneĪs a result, bromination has the greatest utility synthesis of alkyl halide.\) Therefore bromination usually occurs selectively on the most reactive position (the position that forms the most stable carbon radical intermediate), and gives one major product exclusively, as the example here for bromination of isobutane. The relative reaction rate of bromination for different radical is shown here, and you can see the big difference to that of chlorination: Figure 9.4e Relative reaction rate of brominationįor bromination, the reactivity difference between different types of position is so high that the reactivity factor become predominant for determining the product. Bromine is less reactive, means it reactive more slowly, therefore it has chance to differentiate between the different types of hydrogens, and selectively reacts with the most reactive one. The relative lower reactivity of bromine makes it exhibits a much greater selectivity. Instead, bromination with Br 2 can be applied for that purpose. 
Yield % of 1-chloropropane 7.6/13.6 = 56 %īecause of the two major problems for chlorination, lack of selectivity and multi-substitution, chlorination is not useful as a synthesis method to prepare a specific alkyl halide product. The amount of a certain type of product = number of that type of hydrogens × relative reactivityįor the example of monochlorination of propane, the calculation is:Īmount of 1-chloropropane: 6 (number of 1°hydrogens) × 1.0 (relative reactivity) = 6.0Īmount of 2-chloropropane: 2 (number of 2°hydrogens) × 3.8 (relative reactivity) = 7.6 So the overall amount of each isomeric product should be estimated by accounting for both reactivity and probability, that is: With more hydrogen atoms available, the chance for that type of hydrogen to react is higher statistically. Probability simply depends how many hydrogen atoms are there for each type. The relative reaction rate of alkyl radicals for chlorination have been measured and has the approximate values of: Figure 9.4b Relative reaction rate of alkyl radicals for chlorination It has been discussed in section 9.3, that different radicals (primary, secondary or tertiary) have different stability and reactivity. To predict the relative amount of different chlorination product, we need to consider two factors at the same time: reactivity and probability. 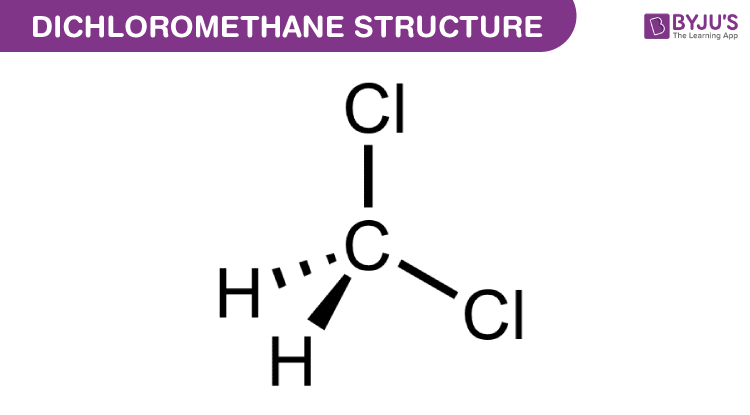
The experimental results of the monochlorination of propane indicate that 45% primary chloride (1-chloropropane) and 55% secondary chloride (2-chloropropane) are produced. As a result, for the reaction of alkane with different hydrogen atoms, a mixture of isomeric monochlorinated products are obtained. Since chlorine is a rather reactive reagent, it shows relative low selectivity, that means Cl 2 does not discriminate greatly among the different types of hydrogens atoms (primary, secondary or tertiary) in an alkane. \)įirst we will focus on monochlorination product, by assuming that chlorination only occur once. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
